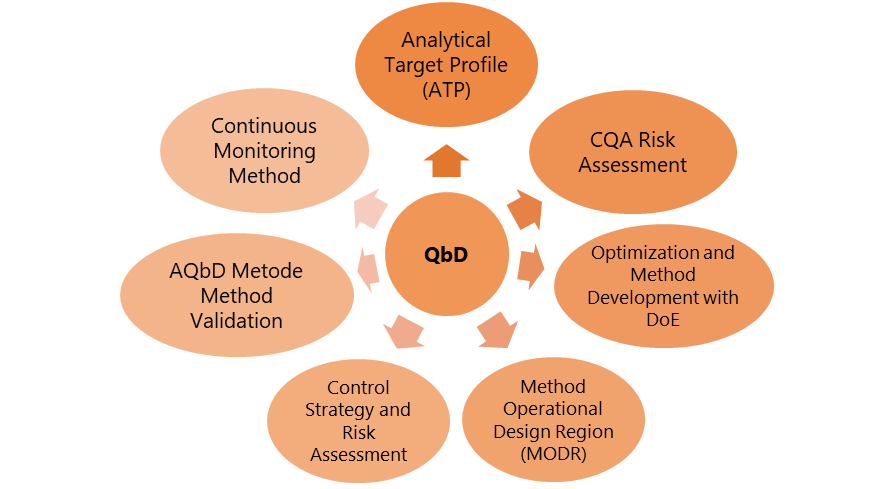
Figure 1 from Analytical Quality by Design Approach to Test Method Development and Validation in Drug Substance Manufacturing | Semantic Scholar
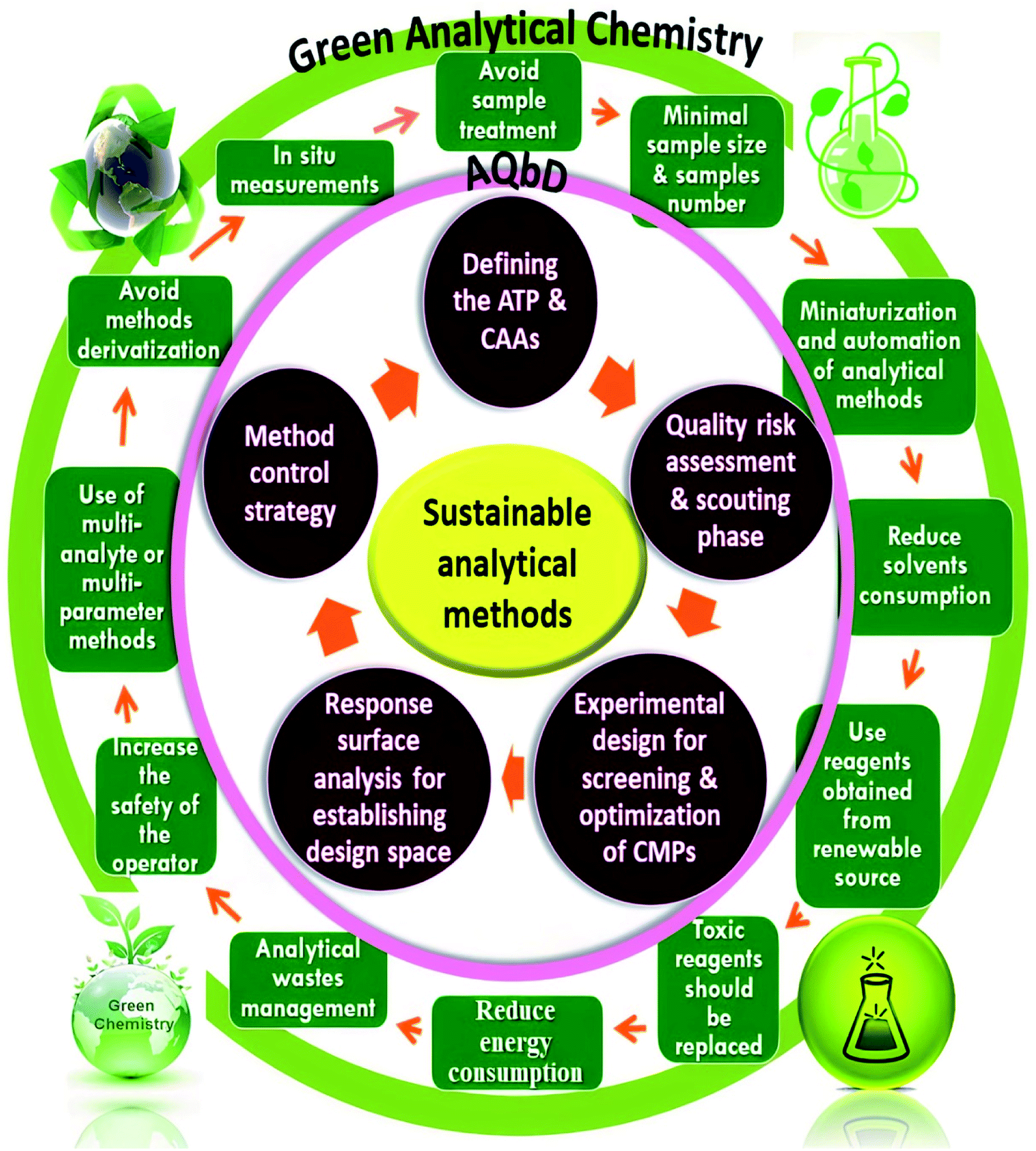
Quality by design approach for green HPLC method development for simultaneous analysis of two thalassemia drugs in biological fluid with pharmacokinet ... - RSC Advances (RSC Publishing) DOI:10.1039/D2RA00966H
Analytical method development by using QbD - An emerging approach for robust analytical method development
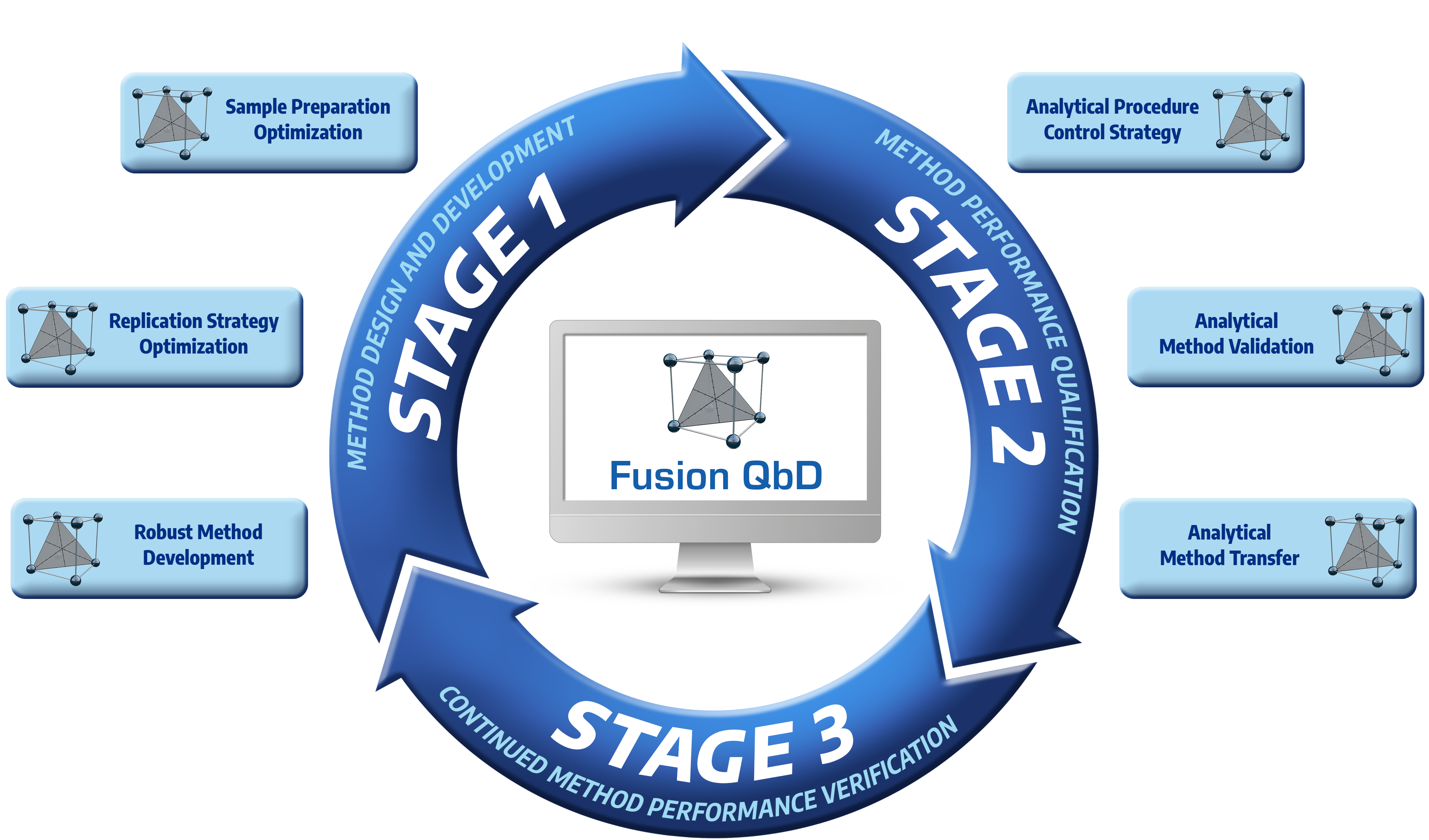
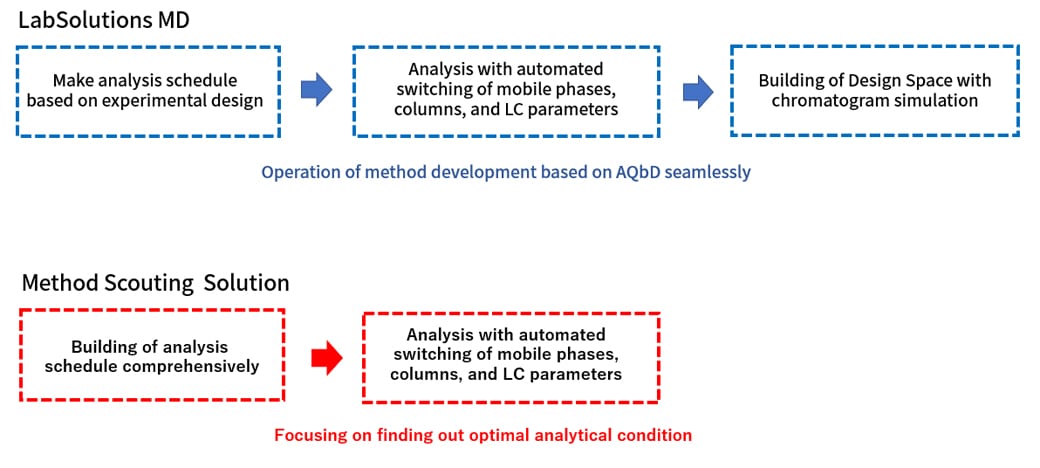
Fusion QbD Software - Quality by Design - DOE - Automated Data Analysis - Chromatography Application Software
![PDF] Analytical Quality by Design Approach to Test Method Development and Validation in Drug Substance Manufacturing | Semantic Scholar PDF] Analytical Quality by Design Approach to Test Method Development and Validation in Drug Substance Manufacturing | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/feb17b38a6e43d7096393c341016bfb3543f64cf/2-Table1-1.png)
PDF] Analytical Quality by Design Approach to Test Method Development and Validation in Drug Substance Manufacturing | Semantic Scholar

Development and Optimization of Liquid Chromatography Analytical Methods by Using AQbD Principles: Overview and Recent Advances | Organic Process Research & Development
Examining the basic principles of quality by design (QbD) approach in analytical studies. - Document - Gale Academic OneFile

Analytical Quality by Design Approach to Test Method Development and Validation in Drug Substance Manufacturing

Development and Optimization of Liquid Chromatography Analytical Methods by Using AQbD Principles: Overview and Recent Advances | Organic Process Research & Development

Analytical Quality by Design Based Method Development for the Analysis of Valsartan and Nitrosamines Impurities Using UPLC-MS | Waters

Analytical Quality by Design Approach to Test Method Development and Validation in Drug Substance Manufacturing

Development and Optimization of Liquid Chromatography Analytical Methods by Using AQbD Principles: Overview and Recent Advances | Organic Process Research & Development